Patient Support Line 1-844-638-7222, Monday-Friday, 8:00 AM – 8:00 PM ET, excluding holidays.

Proven
results with
LUTATHERA
LUTATHERA slowed NET progression for longer in people with newly diagnosed, faster-growing NETs
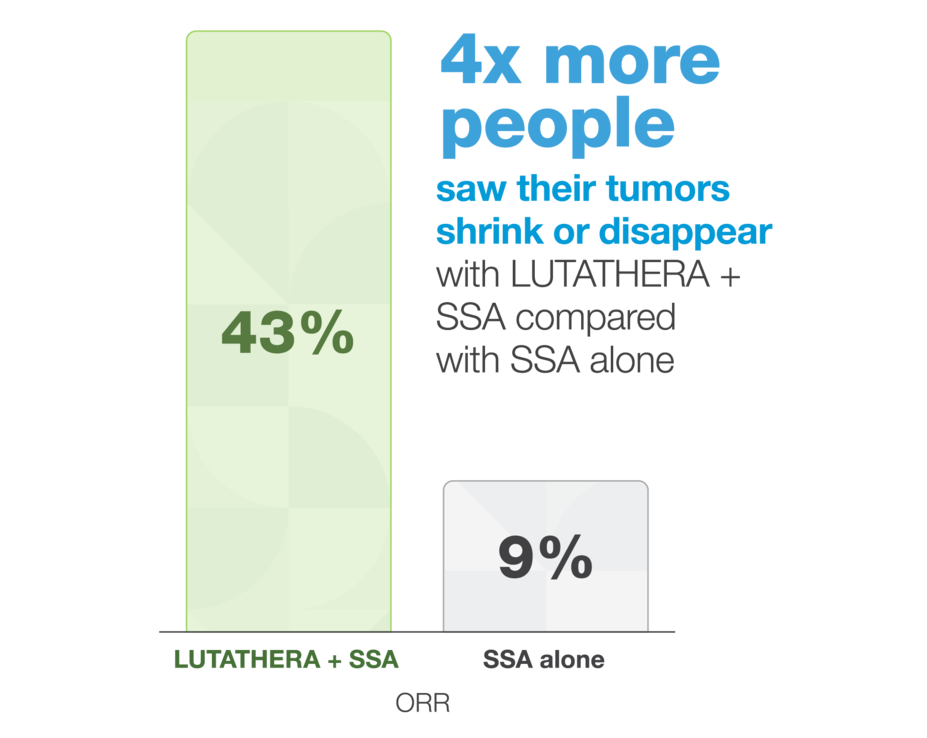
The NETTER-2 trial included 226 people who were newly diagnosed with SSTR+ GEP-NETs that were faster growing (Ki-67, 10%–55%). They were split into 2 groups: 151 received LUTATHERA and a long-acting SSA, and 75 received a long-acting, high-dose SSA alone.
The study measured progression-free survival (PFS). PFS is the amount of time cancer doesn’t grow or spread, also known as progression, during and after treatment. Median PFS is the length of time when half of the people treated have not yet progressed. A longer median PFS means that there is more time before the cancer progresses.
LUTATHERA works with SSA to give people more than 1 year without progression
Tumors were more likely to shrink
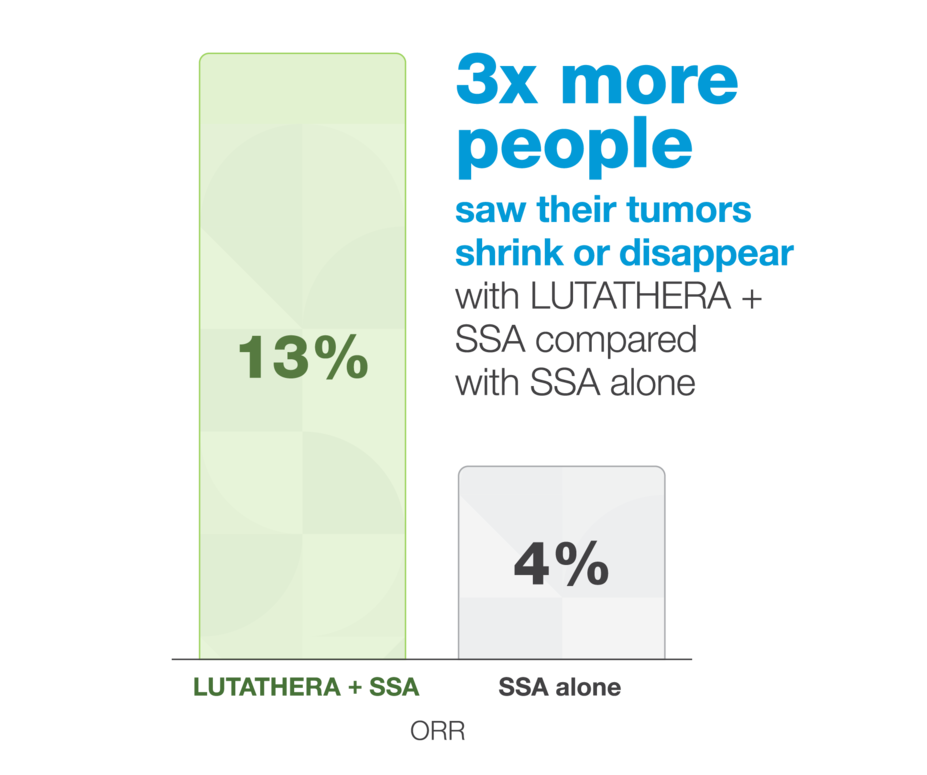
The study also measured objective response rate (ORR). ORR is the percentage of people whose cancer got smaller or disappeared.
LUTATHERA slowed NET progression for longer in people with NETs that progressed after SSA treatment
The NETTER-1 trial included 229 people with SSTR+ GEP-NETs who had tumors that were slower growing (Ki-67, 0%–20%) and progressed on treatment with an SSA. They were split into 2 groups: 116 received LUTATHERA and a long-acting SSA, and 113 received a long-acting, high-dose SSA alone.
If your cancer has progressed after taking an SSA, LUTATHERA with SSA can give you more time without progression
In people taking LUTATHERA + SSA, more than half were progression free at a 14-month check-in. In people taking SSA alone, half had their disease progress at 8.5 months.
Tumors were more likely to shrink with LUTATHERA
GEP-NETs, gastroenteropancreatic neuroendocrine tumors; NET, neuroendocrine tumor; SSA, somatostatin analogue; SSTR+, somatostatin receptor-positive.